INTRODUCTION
Weed infestation remains one of the major constraints to global rice production, causing huge yield losses and increasing the cost of crop management (Kumar et al., 2017). Modern rice-cultivating systems have relied on chemical herbicides for a long time as the primary tool for effective weed management (Pervaiz et al., 2024; Gage et al., 2019). However, the overuse of herbicides has led to the rapid evolution of herbicide-resistant weeds (Beckie, 2006), particularly resistance to acetolactate synthase (ALS)inhibiting herbicides (Saari et al., 2018), which represent one of the most widely used herbicide groups. Among these resistant weeds, Monochoria vaginalis (Burm.f) Kunth (Pontederiaceae), an ever-troublesome broadleaf aquatic weed in paddy fields (Cheng et al., 2015; Hazrati et al., 2023), has increasingly exhibited resistance to ALS inhibitors in many countries including Korea (Hwang et al., 2001; Kuk et al., 2003; Park et al., 2011; Heap, 2025), thereby posing a considerable challenge to weed management programs and threatening the sustainability of rice production systems globally (Islam and Monjardino, 2025).
The growing incidence of herbicide resistance underscores the urgent need for sustainable and environmentally compatible weed control strategies. In this context, reducing reliance on chemical herbicides and adopting integrated weed management strategies are increasingly emphasized as key components of sustainable weed management. Biological control using plant-pathogenic fungi, commonly referred to as mycoherbicides, has been recognized as a promising alternative to chemical herbicides (El-Sayed, 2005). Mycoherbicides offer several advantages, including host specificity, reduced ecological impact, and suitability for integration into eco-friendly agricultural practices (Chakraborty and Ray, 2021). Over the past decades, numerous fungal pathogens have been identified with the potential to selectively control weed species (Weaver et al., 2007), while the potential risk to crop plants still needs to be evaluated under various conditions.
Sclerotium rolfsii Sacc. (synonym: Athelia rolfsii (Cruzi) Tu and Kimbrough), a soil-borne phytopathogenic fungus with a broad host range (Keloth et al., 2024), has recently drawn attention for its potential herbicidal activity against paddy weeds (Tang et al., 2011), particularly M. vaginalis, and also demonstrated its ability to infect and suppress this species (Han et al., 2023; Koo et al., 2023). However, the broad pathogenicity spectrum of S. rolfsii on crops (Yaqub and Shahzad, 2005; Ali et al., 2020) has raised concerns regarding its safety. Before advancing S. rolfsii toward practical field application as a mycoherbicide in the paddy system, a critical step is to assess its selectivity and phytotoxic risk to rice across relevant growth stages, as certain fungal species, such as Sclerotium oryzae Catt., are known to cause stem rot in rice (Prameela et al., 2018). Additionally, evaluating its efficacy against key target weeds including herbicide-resistant biotypes, is essential for determining its suitability and practical value in integrated weed management programs.
Despite the emerging interest in S. rolfsii-based biocontrol, systematic assessments of its safety and weed control performance in rice production systems remain limited. Little is known about its interactions with rice at different growth stages, nor its suppression capacity against ALS-inhibitor-resistant M. vaginalis. Clarifying these unknowns is essential for determining whether S. rolfsii has a potential to serve as a selective mycoherbicide compatible with rice cultivation.
In this study, we conducted a series of glasshouse experiments to (i) evaluate the crop safety of S. rolfsii applied to rice plants at various growth stages, and (ii) investigate its efficacy in managing ALS-inhibitor-resistant M. vaginalis growing with rice to mimic paddy field condition. By integrating phytotoxicity assessment with efficacy evaluation, this research provides valuable evidence supporting the potential of S. rolfsii as a safe and effective mycoherbicide for rice-based weed management systems.
MATERIALS AND METHODS
Two tests were carried out in the glasshouse located at the Experimental Farm Station of Seoul National University in Suwon, Korea (37°27′05″ N, 126°98′90″ E), from 2024 to 2025, to assess (1) the sensitivity of rice to S. rolfsii and (2) the selectivity of S. rolfsii between rice and ALS-inhibitor-resistant M. vaginalis growing together.
Test 1: Rice sensitivity test to S. rolfsii
Rice (Oryza sativa L., cv. Chucheong) was used as a test crop. Seeds were germinated and then incubated to reach rice seedling 2-leaf stage in the growth chamber (Hanbaek Scientific Co., Korea) at 30/20°C (day/night). Rice seedlings were transplanted into pots (15 cm × 11 cm × 8.5 cm) filled with mixed soil consisting of paddy soil and commercial horticultural soil (Baroker, Seoul Bio, Korea) at a volumetric ratio of 2:1. To obtain plants at different developmental stages (3-, 5-, and 7-leaf stages), transplanting was conducted at three time points at one-week intervals. Planting density was standardized at five seedlings per row and eight seedlings per column. Pots were placed and submerged in large trays (57 cm × 36 cm × 14 cm) and maintained under glasshouse conditions (35/25°C, day/night) until the plants reached the intended leaf stages.
A total of 36 pots containing rice plants were used, with 12 pots assigned to each leaf stage. Irrigation water was completely drained from the pots one day prior to treatment. Sclerotia of S. rolfsii (0.05–0.1 cm in diameter) were then applied evenly to the soil surface at four dose rates, 0, 20, 40, 80 kg sclerotia ha-¹. Each treatment was replicated three times, and pots were arranged according to the randomized complete block design in the glasshouse.
Plants were visually assessed at 10-day intervals for infection symptom development and RGB images were captured periodically. For final assessment, plant height, tiller number, and fresh weight of rice were measured at 30 days after treatment (DAT).
Test 2: Selectivity test of S. rolfsii between rice and ALS-inhibitor-resistant M. vaginalis
Rice (Oryza sativa L., cv. Chucheong) and ALS-inhibitor-resistant M. vaginalis, collected from rice fields where ALS-inhibiting herbicides had failed to control (Lim et al., 2018), were used in the selectivity test. Due to differences in growth rates between the two species, ALS-inhibitor-resistant M. vaginalis seedlings were established earlier than rice.
Rice seedlings were prepared as described in Test 1 and transplanted into large square trays (38 cm x 24 cm x 6 cm) arranged in two rows with four hills per row and three seedlings per hill. Subsequently, ALS-inhibitor-resistant M. vaginalis seedlings at the 3-leaf stage were transplanted into the same trays at a density of 50 plants per tray and maintained under glasshouse conditions until reaching the 7-leaf stage.
A total of 24 trays containing co-cultivated rice and M. vaginalis plants were used in this test. Irrigation was withheld one day prior to treatment and then sclerotia of S. rolfsii were applied uniformly to the soil surface at five dose levels including an untreated control and four application doses corresponding to 10, 20, 40, and 80 kg sclerotia ha-¹. In addition, a commercial herbicide, penoxsulam (SalchoDaecheop™, Corteva Agriscience), which is highly effective for broad-leaf and sedge weeds control, was applied as a foliar spray at the recommended rate of 30 g a.i. ha-¹ as a positive control. Treatments were replicated four times and arranged in a randomized complete block design in the glasshouse. Sub-irrigation was made if necessary.
Visual assessments were conducted to evaluate the extent of plant damage at 10 and 20 DAT. RGB image acquisition and fresh weight measurements were performed at 20 DAT.
Statistical analysis
For ALS-inhibitor-resistant M. vaginalis, shoot fresh weight was normalized and expressed as a percentage of the untreated control, while original observed data were used for rice growth and visual assessments. All the data were subjected to analysis of variance (ANOVA) using Prism version 7.04 (GraphPad Software, USA) and SAS® version 9.4 (Statistical Analysis System, USA). When the ANOVA indicated significant treatment effects, mean comparisons were performed using Fisher’s least significant difference (LSD) test at a significance level of α = 0.05.
Results and Discussion
Sensitivity of rice to S. rolfsii at various rice growth stages
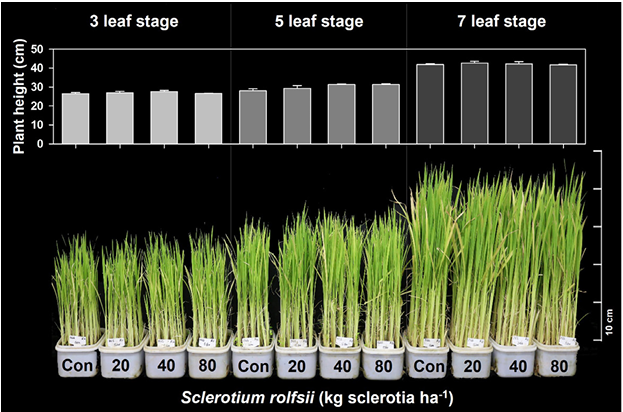
Visual assessment revealed that rice plants exhibited no observable phytotoxic effects following application of S. rolfsii across all tested leaf stages and dose levels (Fig. 1). At the 3-, 5-, and 7-leaf stages, rice plants treated with sclerotia of S. rolfsii at 20, 40, and 80 kg sclerotia ha-1 displayed statistically similar shoot height, canopy density, and overall growth compared to those of the untreated control. Plant height ranged from 26.50–27.62, 27.96–31.33, and 41.74–42.66 cm, at the 3-, 5-, and 7-leaf stages, respectively, with no significant differences among S. rolfsii application rates within each rice growth stage. No visible pathogenic symptoms, chlorosis, necrosis, or growth suppression were detected at any assessment time point. Increasing application rates did not result in rice damage even at the highest dose of 80 kg sclerotia ha-1. Rice plants maintained normal growth regardless of rice growth stage and dose of sclerotia application, indicating a high level of tolerance to S. rolfsii irrespective of application timing. These observations demonstrate that S. rolfsii did not adversely affect rice growth under the experimental conditions used in this study.
The absence of phytotoxic effects across multiple growth stages suggests that rice possesses a strong inherent tolerance to S. rolfsii. This tolerance may be partly explained by the host specificity of the pathogen, as S. rolfsii is most associated with legumes, crucifers, and cucurbits (Javaid et al., 2020) and appears to have limited infection efficiency on rice tissues, or effective defense responses that restrict fungal establishment. Unlike certain pathogenic fungi reported to cause rice diseases (Conde et al., 2025), S. rolfsii did not induce stem rot-like symptoms or growth inhibition, even under high air temperature (27-35℃) and humidity conditions favorable for sclerotial development (Kator et al., 2015).
From an application standpoint, the consistent lack of damage across leaf stages indicates a wide safety margin for rice, thereby reducing concerns related to precise application timing. This characteristic is particularly advantageous for mycoherbicide development, as it enhances operational flexibility under field conditions where crop growth stages may vary. Overall, these results provide strong evidence that S. rolfsii is non-phytotoxic to rice across a broad range of growth stages and application rates, supporting its potential use as a selective mycoherbicide in paddy systems when targeting susceptible weed species.
Selectivity of S. rolfsii between rice and ALS-inhibitor-resistant M. vaginalis
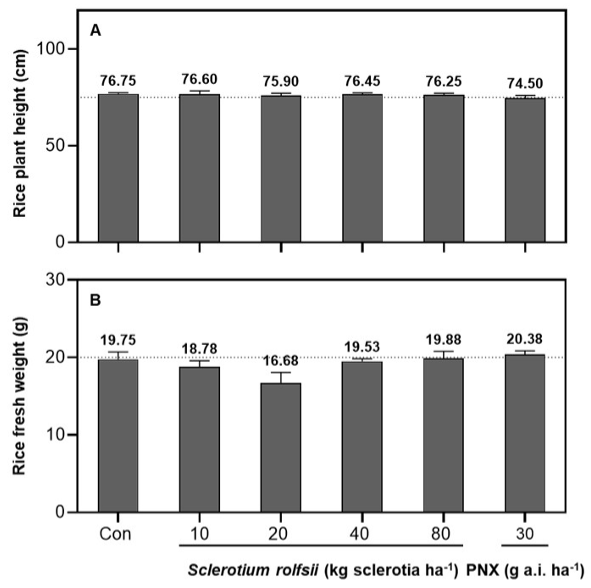
At 20 DAT, application of S. rolfsii at a range of doses (10-80 kg sclerotia ha-1) showed no significant phytotoxic effects on rice growth. Rice plant height and fresh weight did not differ significantly between S. rolfsii treatments and the untreated control (p > 0.05), indicating good rice safety (Fig. 2, Fig. 3A, 3B). In contrast, ALS-inhibitor-resistant M. vaginalis was significantly suppressed by S. rolfsii in a dose-dependent way (Fig. 4). Weed control efficacy based on visual growth reduction increased progressively with increasing application dose, reaching approximately 84.25%, 86.25%, 75.50%, and 93.50% control at 10, 20, 40, and 80 kg sclerotia ha-¹, respectively, at 20 DAT (Fig. 4A). Such significant growth reduction resulted in significant fresh weight reduction (Fig. 4B). Even at the recommended dose of S. rolfsii (20 kg sclerotia ha-1), the fresh weight of M. vaginalis was reduced to 27.06% of the untreated control, indicating strong weed suppression. The chemical herbicide penoxsulam (30 g a.i. ha-¹) provided limited suppression of M. vaginalis, with only about 22.5% control, so failed to control the weed, confirming that M. vaginalis is penoxulam resistant, i.e., ALS-inhibitor-resistant. Overall, S. rolfsii treatments, particularly at dose ≥20 kg sclerotia ha-¹, demonstrated greater weed control efficacy, suggesting its potential for the management of ALS-inhibitor-resistant M. vaginalis whose control is failed by ALS-inhibitor-based herbicide systems including penoxsulam.
Fig. 4
Visual growth reduction (A) and fresh weight (B) of Monochoria vaginalis at 20 days after treatment of Sclerotium rolfsii at a range of doses and penoxsulam (PNX) at 30 g a.i. ha-1. Error bars represent the mean ± SEM (n = 4). The vertical bars represent least significance difference (LSD) at p < 0.05.

In summary, our results clearly demonstrate that S. rolfsii functions as a selective mycoherbicide, exhibiting high rice safety as well as sufficient efficacy against ALS-inhibitor-resistant M. vaginalis. The absence of pathogenic symptoms and growth suppression in rice at various growth stages from the 3-leaf stage by S. rolfsii even at 80 kg sclerotia ha-1 (x 4 of the recommended dose) confirms that S. rolfsii is selectively safe for paddy rice. In contrast, M. vaginalis showed high susceptibility to S. rolfsii, with weed control efficacy increasing with dose. The observed dose-response relationship suggests that higher inoculum densities enhance pathogenhost interactions, leading to greater infection development and biomass reduction in the target weed. This pattern is consistent with previous findings that effective colonization and infection by mycoherbicides are closely related to application dose and propagule density (Kator et al., 2015).
Notably, S. rolfsii outperformed the chemical herbicide penoxsulam under the same experimental conditions. While penoxsulam failed to control M. vaginalis due to its resistance to ALS-inhibitors the mycoherbicide achieved up to 83.97% control, highlighting its superior efficacy against ALS-inhibitor-resistant M. vaginalis, suggesting that S. rolfsii may also be used to manage the other broadleaf weeds, whose control was failed by ALS-inhibitor herbicides. Our results suggest that S. rolfsii has strong potential as an alternative or complementary weed management tool, particularly in situations where chemical herbicides show reduced effectiveness or resistance issues (Aneja et al., 2017). Overall, these findings indicate that S. rolfsii is a promising mycoherbicide candidate for the management of ALS-inhibitor-resistant M. vaginalis in rice paddies. Further studies on sclerotial characteristics, such as germination and mycelial growth, are needed to improve the practical use of S. rolfsii as a mycoherbicide.
요약
미생물제초제는 증가하는 제초제 저항성 잡초에 대응하기 위한 화학 제초제의 대안으로 주목받고 있다. 그 중 Sclerotium rolfsii는 여러 잡초종에 대해 제초활성을 보이는 것으로 보고되었으나, 작물 재배 체계에서의 안전성에 대해서는 아직 충분히 규명되지 않았다. 본 연구는 S. rolfsii의 벼에 대한 안전성과 아세토락테이트 합성효소(ALS) 저해제 저항성 물달개비에 대한 방제 효과를 평가하고자 수행되었다. 벼 감수성 평가에서 S. rolfsii는 벼의 3-, 5- 및 7- 엽기에 처리되었다. 약제 처리 후 30일 동안 벼에서 약해나 균사의 형성은 관찰되지 않았으며, 초장과 생체중 또한 엽기 및 처리 약량에 따라 유의한 차이를 보이지 않았다. 이러한 결과를 통해 S. rolfsii가 벼에 대한 병원성을 나타내지 않음을 확인하였다. 또한 벼 재배 포장에서의 물달개비 방제 시의 벼에 대한 미생물제초제의 안전성을 평가하기 위해 벼와 물달개비 동시 재배 조건에서의 평가를 수행하였다. 약제 처리 후 20일에 벼에서는 생육 억제가 관찰되지 않은 반면, 물달개비에서는 현저한 생육 억제가 나타났으며, 모든 S. rolfsii 처리구에서 69.94%~83.97%의 방제 효과를 보였다. 결론적으로 S. rolfsii의 벼에 대해 높은 안전성과 ALS 저해제 저항성 물달개비에 대해 우수한 제초 활성은 벼 재배에서 제초제 저항성 물달개비의 방제관리를 위한 미생물제초제로서의 활용 가능성을 시사하였다.
Acknowledgements
This research was supported by the Korea Institute of Planning and Evaluation for Technology in Food, Agriculture and Forestry (IPET) Ministry for Food, Agriculture, Forestry and Fisheries, Republic of Korea (Project No. 321056-05-1-HD050). The authors also thank Moghu Research Center, Korea for providing the research materials (Sclerotium rolfsii BWC 98-105).